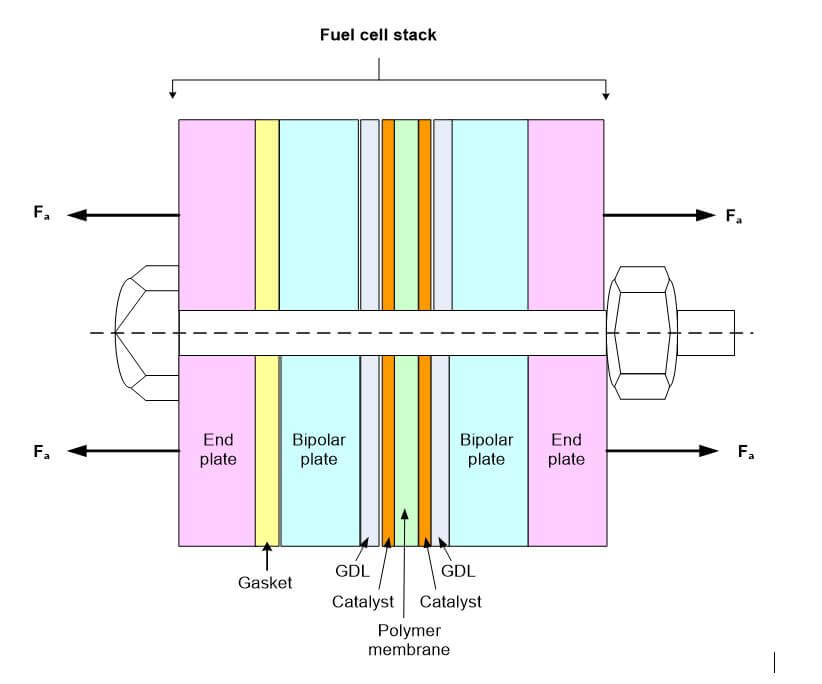
There are many steps involved in the manufacturing of a fuel cell stack. One of these steps is the hot pressing of the polymer electrolyte membrane to the two gas diffusion layers (GDLs). This creates a three-layer laminate membrane electrode assembly (MEA). Other steps involve the machining or etching of the end plates, bipolar plates and cooling plates, and the sizing of the gaskets, contacts and MEA surrounds. After all of the fuel cell components have been manufactured, they are stacked together and clamped using a clamping mechanism such as bolts. The contact resistance, mass and charge transfer between the electrolyte membrane and the GDL is very good due to the fusion of the three layers. In contrast, the remaining layers are separated until they are clamped together using bolts or some other type of clamping device. Therefore, the interfacial resistances between the remaining layers are significant. The contact, cooling and bipolar plates are clamped together, and since the cooling and contact layers are similar, and the surface roughness can be considered negligible, the contact resistance between these layers is small when the stack is clamped together. The interface that is most affected by the clamping pressure is the GDL and bipolar plate interface. The material properties of these adjoining layers are extremely different, and since the GDL layer is porous, it is highly sensitive to the clamping pressure. Not only does the GDL thickness change with clamping pressure, but the change in thickness also affects the porosity and permeability of the GDL. The bolt torque, clamping force, contact resistance and permeability of the GDL all affect the electrochemical performance of a PEM fuel cell by influencing the ohmic and mass-transport polarizations inside of the fuel cell.
Materials bolted together withstand moment loads by clamping the surfaces together, where the edge of the part acts as a fulcrum, and the bolt acts as a force to resist the moment created by an external force or moment. Figure 1 shows forces exerted by the clamped materials (fuel cell layers) on a clamping bolt and nut. The forces exerted by the tightening bolts are due to the bolt material properties, the properties of the materials being clamped together, and the torque applied to the bolts.
Figure 1. The forces exerted by the clamped materials (fuel cell layers) on the bolt and nut.
Materials that are held together by a bolt are subjected to a force a distance away from the center of stiffness of a bolt pattern. In order to determine the optimal torque for a bolt, a maximum bolt force can be calculated based upon the maximum amount of stress and force that can be applied to the bolt, and the “joint” which is the fuel cell stack in this case. The optimal torque can be found by calculating the force that can be applied to joint until the force on the joint is lost. Assuming each of N bolts is a distance from the bolt pattern’s center of stiffness, each bolt has the same force and there is a coefficient of friction between the bolted members.
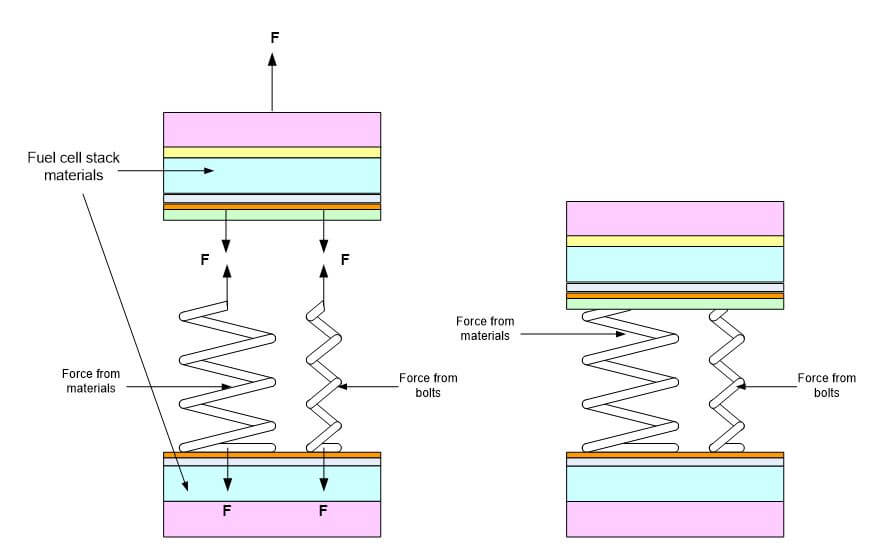
Tightening the bolts stretches the bolts and compresses the stack materials. If an external force is applied to the stack, the optimal torque usually means that the stack stays compressed. This ensures proper stiffness and fatigue life of the stack. Figure 2 shows how the region under a bolt head acts like a spring.
Figure 2. The forces exerted by the clamped materials and bolt.
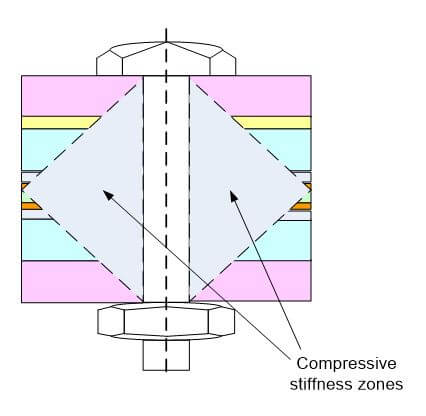
Of the energy created by the bolt force, about 50% of the energy goes to friction under the bolt head, 40% goes to friction in the threads and about 10% goes to create tension in the threads. The rotation of the bolt head relative to the parts being bolted together is a good measure of tension in the bolt. As shown in Figure 3, there is a strain or stress cone under the bolt head that project from 30 to 45 degrees from the vertical, and 45º is most commonly used for bolt torque calculations.
Figure 3. Compressive stiffness zones underneath a bolt head in a fuel cell stack.
When determining a fuel cell stack design and the optimal bolt torque, two questions need to be answered:
1. How much tension does the bolt actually create?
2. What is the ideal tightness?
In bolt science, the optimal torque can be found by calculating the forces that the bolt can withstand, along with the stiffness of the materials being clamped, and the desired tightness that the clamped materials require. In a fuel cell stack, the bolt material can handle more force than the fuel cell stack needs for optimal performance. Therefore, to find the ideal torque for a fuel cell stack, the effects of compression have to be considered.
The contact resistance and GDL permeability is governed by the material properties of the contacting GDL and bipolar plate layers. The contact resistance is most reliant on the contact between these layers. The contact resistance between the catalyst and membrane layers is low because they are fused together, and the contact resistance between the bipolar plates and other layers is low because the materials are typically non-porous with similar material properties (high density, with similar Poissons’ ratios and Young’s Moduli). The GDL and the bipolar plate layers have several characteristics that make the contact resistance and permeability larger than between the other layers:
1. The material properties are very different (Poissons’ ratios and Young’s Moduli) - (a hard material with a soft material)
2. The GDL layer is porous, and the permeability has been reduced due to the reduction in pore volume or porosity, and
3. Part of the GDL layer blocks the flow channels that are in the bipolar plate creating less permeability through the GDL as the compression increases.
As shown in Figure 4, both the in-plane and through-plane conductivities increase as the compressed thickness of the GDL was decreased. The conductivities have a linear dependence on the GDL compressed thickness. This may be due to the reduced porosity of the GDL, which leads to shorter distances between conductive carbon fibers and better contacts between the fibers.
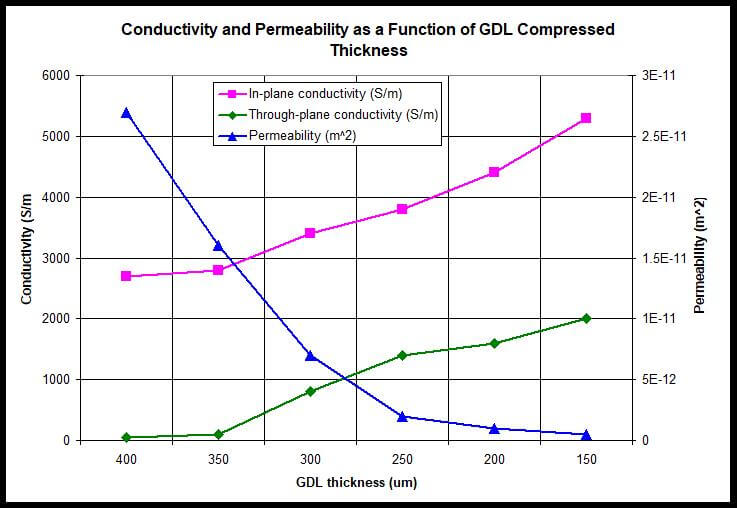
Figure 4. Conductivity and Permeability as a Function of GDL Compressed Thickness.
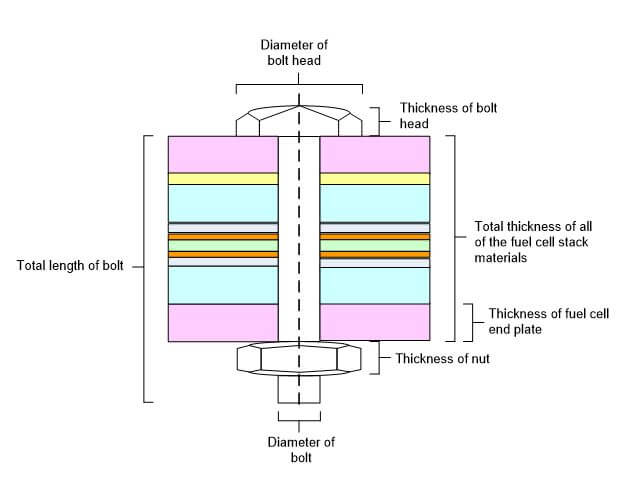
In order to accurately determine the ideal clamping pressure (tightening torque) for a fuel cell stack, the stiffness of the materials between the bolts can be estimated. The stiffness of the materials includes the compressive stiffness of the materials under the bolt head in series with the stiffness of the physical interface, which increases with pressure, and the stiffness of the threaded material. Some of the dimensions that can be used in the bolt and layer stiffness calculations are shown in Figure 5.
Figure 5. Dimensions Used in the Bolt and Layer Stiffness Calculations.
Figure 6 shows actual polarization curves of a PEM fuel cell under five different clamping pressures. The current is dynamically stable for four of the five clamping pressures. The lowest clamping pressure of 28 oz-in displayed the worst I-V performance, due to mass transfer limitations and higher contact resistance. The polarization curves continuously increase until a torque of 36 oz-in is reached. As the torque continues to increase to 44 oz-in, the polarization curves again begin to decrease.
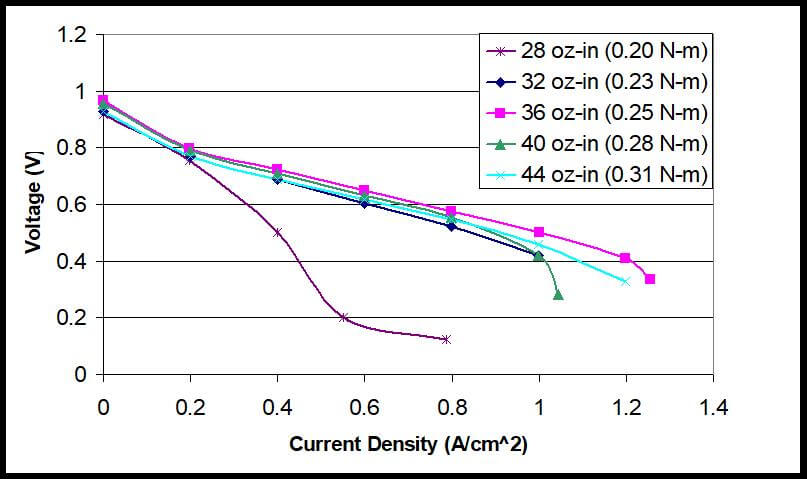
Figure 6. Polarization Curves with Tightening Torques of 28 oz-in to 44 oz-in for a fuel cell stack.
Figure 7 displays the performance curves for another fuel cell stack with five different clamping pressures. Again, the polarization curves reflect the effect of the interfacial electrical resistance, mass transfer and optimal clamping pressure on the fuel cell stack. As seen in Figure 7, the fuel cell performance appears to be the poorest with the 6 oz-in clamping pressure. Compression with a torque of 10 oz-in shows the best performance curve. As the torque increased from 10 to 14 oz-in, the fuel cell performance decreased as the mass transfer is hindered due to the decreased porosity of the GDL layer.
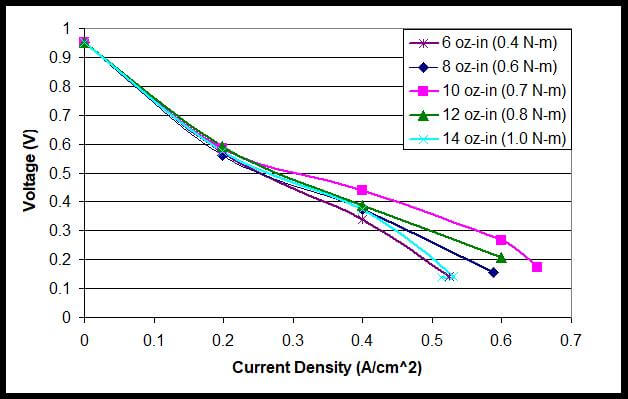
Figure 7. Polarization Curves with Tightening Torques of 6 oz-in to 14 oz-in for a Fuel Cell Stack.
As shown in Figure 8, the case of 4 oz-in compression showed the best polarization curve. As with the previous polarization curves for the other fuel cell stacks, the lowest torque showed a poor polarization curve in comparison with the polarization curve obtained with the optimal torque. It seems to be difficult to achieve more than 40 mA cm−2 of current density with a compression of 6 oz-in due to the mass-transfer limitation.
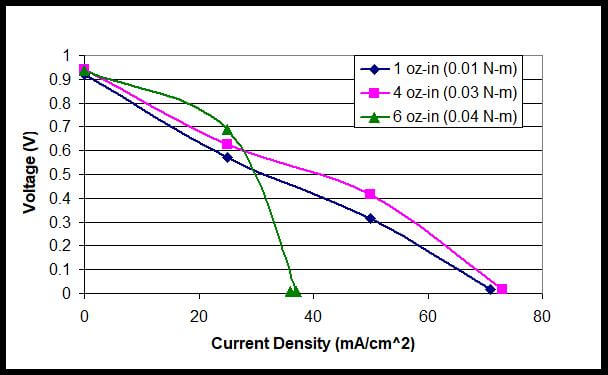
Figure 8. Polarization Curves with Tightening Torques of 1 oz-in to 6 oz-in for a Fuel Cell Stack.
The torque is used as an indirect means of measuring the stack clamping pressure, which has a direct effect on fuel cell stack performance. We experimentally examined the effect of the clamping pressure on the electro-physical properties on three different free-convection PEM fuel cell stacks. As the stack material stiffness, bolt material, or GDL compression changed, the resulting fuel cell polarization curve changed. We see that low tightening torque results in a high interfacial resistance between the bipolar plate and the gas diffusion layer that reduces the electrochemical performance of a PEM fuel cell. In contrast, high tightening torque reduces the contact resistance between the graphite plate and the gas diffusion layer but narrows down the diffusion path for mass transfer from gas channels to the catalyst layers. We see from our experiments that the correct balance of tightening torque for a certain stack and bolt stiffness, combined with minimal contact resistance and mass transfer limitations within a fuel cell stack can lead to optimal fuel cell performance.

 Posted by
Posted by


















Enter the code in the box below: