Creating high-efficiency fuel cells requires proper temperature control, and heat management to ensure that the fuel cell system runs consistently. Depending upon the fuel cell type, the optimal temperature can range from room temperature to 1000 ºC, and any deviation from the designed temperature range can result in lowered efficiencies. Higher temperatures also mean faster kinetics and a voltage gain that in general exceeds the voltage loss from the negative thermodynamic relationship between the open-circuit voltage and temperature. Lower temperatures mean shorter warm-up times for the system and lower thermomechanical stresses. Corrosion and other time- and temperature-dependent processes are retarded, and much less water is required for the saturation of input gases. In the case of PEMFCs, a higher operating temperature means more of the product water is vaporized; thus, more waste heat goes into the latent heat of vaporization, and less liquid water is left to be pushed out of the fuel cell.
The temperature in a fuel cell is non-homogenous, even when there is a constant mass flow rate in the ducts. This is due to heat transfer, and for certain fuel cell types, a phase change of the reactants or products. To accurately predict temperature-dependent parameters as well as rates of reaction and species transport, the temperature and heat distribution needs to be determined accurately. Convective heat transfer occurs between the solid surface and the gas streams, and conductive heat transfer occurs in the solid and/or porous structures. The reactants, products, and electricity generated are the basic components to consider in modeling basic heat transfer in a fuel cell as shown in Figure 1.
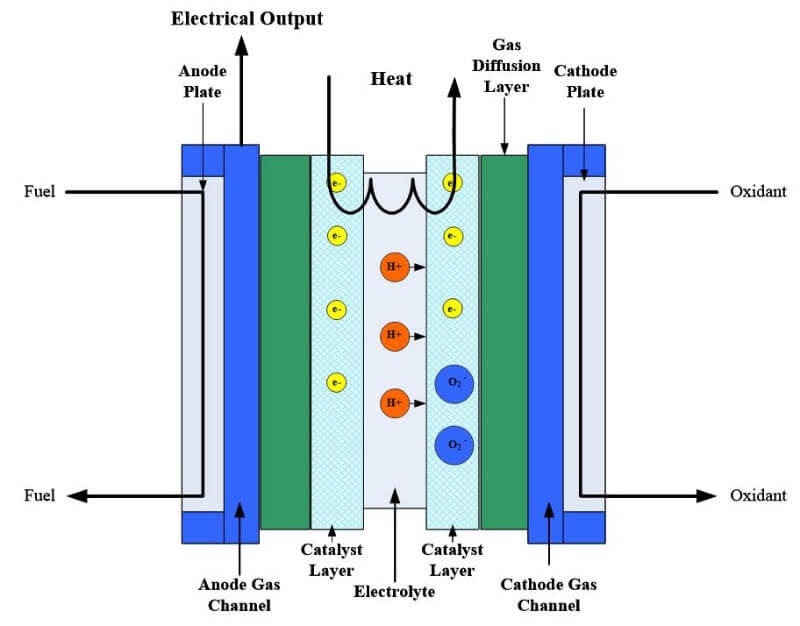
Figure 1: Stack illustration for heat flow study
There are many sophisticated programs to help determine the heat distribution in the fuel cell stack. If you do not have access to one of these programs, many insights regarding the heat distribution can still be made using simple energy balances and tradition heat transfer equations and analysis. The first step in any simple analysis is to complete energy balances. The total energy balance around the fuel cell is based upon the power produced, the fuel cell reactions, and the heat loss that occurs in a fuel cell. The energy balance varies for each fuel cell type because of the different reactions that occur in different fuel cells. The general energy balance states that the enthalpy of the reactants entering the cell equals the enthalpy of the products leaving the cell plus the sum of the heat generated, the power output, and the rate of heat loss to the surroundings.
The fuel cell energy balance is the sum of all the energy inputs and the sum of all the energy outputs:
The inputs are the enthalpies of the fuel, the oxidant, and the water vapor present. The outputs are the electric power produced, enthalpies of the flows out of the fuel cell, and the heat leaving the fuel cell through coolant, convection, or radiation.
The enthalpy (J/s) for each dry gas or mixture of dry gases is
where m is the mass flow rate of the gas or mixture (g/s), Cp is the specific heat (J/[g*K]), and T is the temperature in ºC.
If the gas has a high heating value (combustible), its enthalpy is then:
where hHHV0 is the higher heating value of that gas (J/g) at 0 ºC. The heating values are usually reported at 25 C, therefore, the higher heating value may need to be calculated at the chosen temperature. The enthalpy of water vapor is
The enthalpy of liquid water is
The inputs and outputs of the energy balance can quickly become complicated when the heat balance is performed for each fuel cell layer and/or stack heating and cooling is involved.
Heat generated by the electrochemical reaction in the anode catalyst layer can be given by:
where 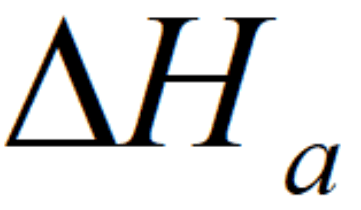
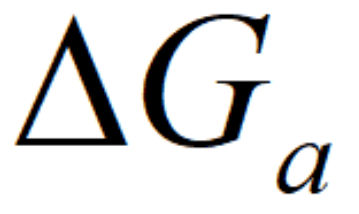
A temperature gradient within a homogenous substance results in an energy transfer through the medium. The rate of heat transfer in the x-direction through a finite cross-sectional area, A, is
where k is the thermal conductivity, W/(m*k). Every material in the fuel cell stack has different levels of thermal conductivity.
There is also heat lost by the stack through natural convection and radiation due to the surroundings. For small stacks or single cell stacks, the surface area is large, and the heat dissipation is greater than the heat generation.
Cooling can be achieved through several means. Passive cooling of the fuel cell, on the other hand, can be achieved with cooling fins and heat sinks. The fuel cell can also be coupled with subsystems that absorb heat, like turbine reheaters and metal hydride containers. In the case of PEMFCs, the evaporation of some product water at the cathode absorbs some heat, while active cooling with air or liquid coolants can be used to transfer heat to radiators. Most fuel cell stacks require a cooling system to maintain temperature homogeneity throughout the fuel cell stack. Portable fuel cells need to maintain the correct temperature despite temperature, humidity, vibration, and impact changes due to the environment. Very small and micro fuel cells may not require a cooling system and often can be designed to be self-cooled.
There are simple fuel cell system designs with cooling plates inserted into the stack or cooling channels machined into the bipolar plates. The coolant is cooled by a heat exchanger and moved through the system using a coolant pump. There are usually thermocouples or temperature sensors in the stack that feed back to a flow controller which adjusts the flow based on the amount of coolant required. There are many cooling methods and designs for every type of fuel cell system. Some of the stack cooling options available are:
• Cooling through free convection (air flow)
• Cooling using a condenser
• Cooling using heat spreaders
• Cooling using cooling plates
There are also many other methods for cooling the stack. The methods presented in this section are the most often used.
One of the simplest solutions for cooling a fuel cell stack is through free convection. This method does not require any complicated designs or coolant and can be suitable for small or low-power fuel cell stacks. Heat dissipation can be achieved through manufacturing fins, or through an open cathode flow field design. This cooling method does not have adequate temperature control because the cooling depends largely on the temperature and humidity of the environment. It is also difficult to recover waste heat for power using free convection.
Condenser cooling allows the stack to operate at higher temperatures than other cooling types. For PEM fuel cells, water can be condensed from the exhaust and then reintroduced into the stack. The condenser must be kept at the water balance temperature so that the overall system will not gain or lose water. Therefore, this system requires precise temperature control like in stack cooling plate systems.
Another option for cooling a stack is to use heat spreaders. Heat can obviously be transferred more efficiently outside the stack. Heat spreaders can help transfer heat to the outside of the stack through conduction, and then the heat can dissipate into the surroundings using natural or forced convection. If this method is chosen, high-performance heat spreaders must be used.
Using cooling plates is a common solution for cooling fuel cell stacks. Thin cooling plates can be manufactured and inserted into the fuel cell, or additional channels can be machined in the bipolar plates to allow air, water, or coolant to flow through the channels to remove heat from the stack. The overall system design can be as simple as only one coolant recirculation path. Precise temperature control is needed to accurately maintain the temperature within a selected range. When cooling plates are used in a PEM fuel cell system, a simultaneous water and heat balance must be conducted to avoid dehydration of the membrane and flooding within the stack.
The heat transfer and total energy balance around the fuel cell are based upon the power produced, the fuel cell reactions, and the heat loss that occurs in a fuel cell. The energy balance varies for each fuel cell type because of the different reactions that occur in different fuel cells. The general energy balance states that the enthalpy of the reactants entering the cell equals the enthalpy of the products leaving the cell plus the sum of heat generated, the power output, and the rate of heat loss to the surroundings. After the initial energy balance equations have been calculated, more sophisticated heat transfer modeling software can be used to more accurately product the heat flow through the stack. These predictions are very useful when designing the heat management system for your fuel cell stack.

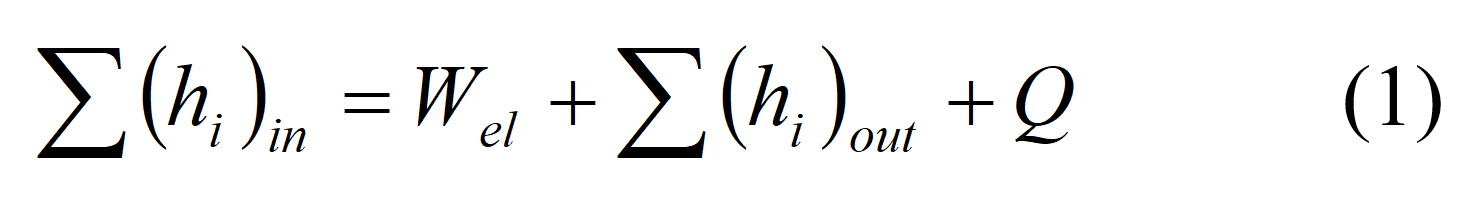

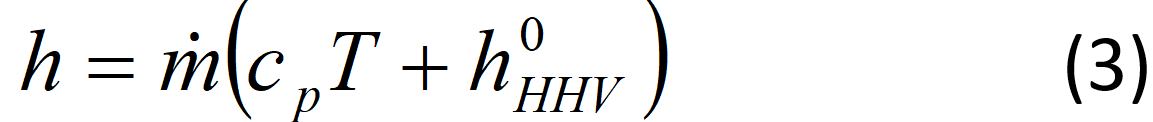
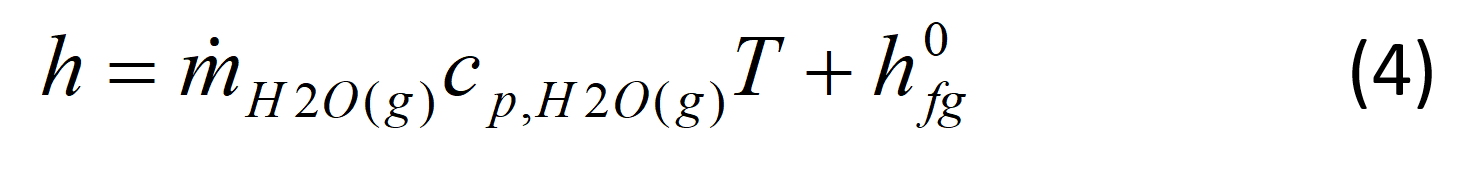
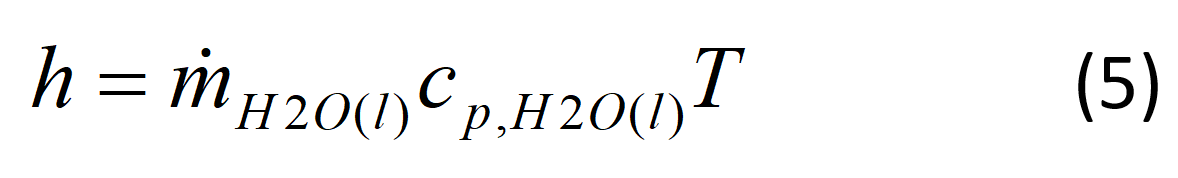
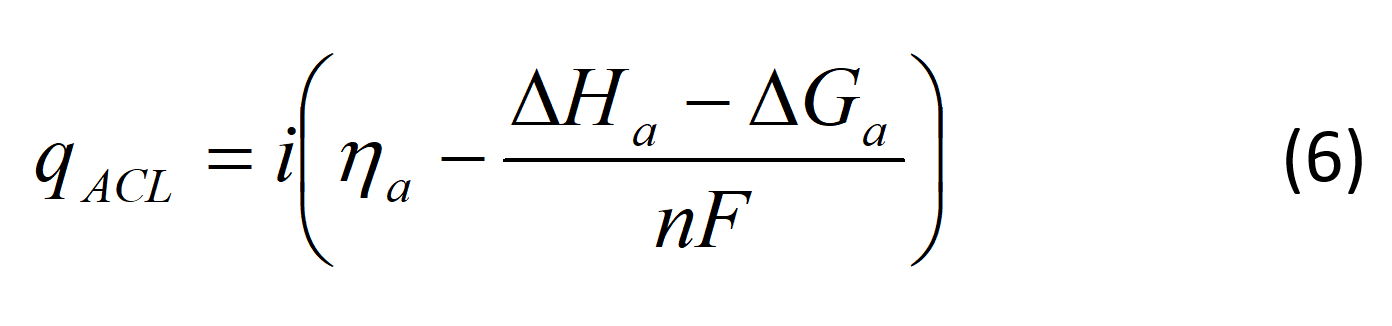
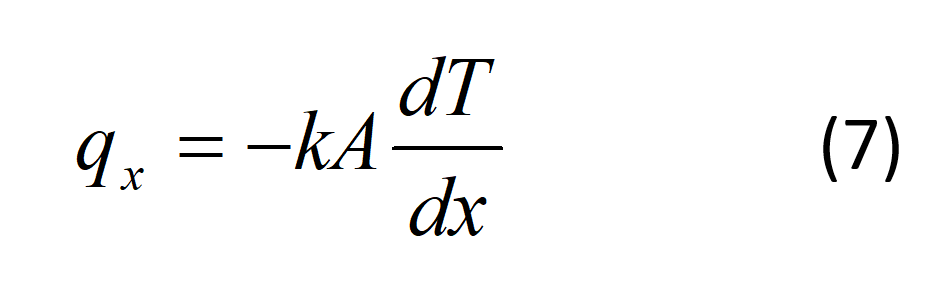
 Posted by
Posted by






























Enter the code in the box below: