Ion exchange materials are used to purify, separate, and extract many different types of molecules, including organic and biochemical molecules. When ion exchange materials involve these ion types, there may be additional complexities involved with the interaction. Some of the phenomena that may occur are:
- Secondary forces between the ionized group and counterion. These forces may consist of coordination, hydrogen, and van der Waals bonding.
- The pH can affect the percent ionization.
- The position of the functional groups can affect ion transport.
- Hydration of organic molecules can be more complex than inorganic ions.
- Organic ions may be larger than inorganic ions; thus, steric hinderances can reduce ionic interactions.
Therefore, ion exchange phenomena may be able to be explained chemically by stoichiometric reactions, but the actual ionic selectively may be determined by other interactions.
Effect of Coordination, Hydrogen, and van der Waals Bonds
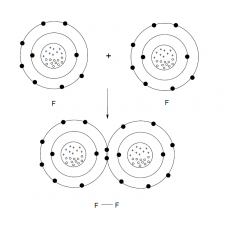
There are several types of forces that can contribute to the total strength of the ionic interaction. A few of these forces were discussed in your introductory chemistry classes, such as coordination, hydrogen, and van der Waals forces. Recall from chemistry that a covalent bond occurs when elements share electrons to obtain a full outer shell (octet). For example, a fluorine atom has seven valence electrons (F is in Group VII on the Periodic Table). Therefore, each fluorine needs one electron to obtain an octet (8 electrons in the outer shell), and each fluorine needs to share one electron. Figure 1 illustrates the F-F covalent bonding.
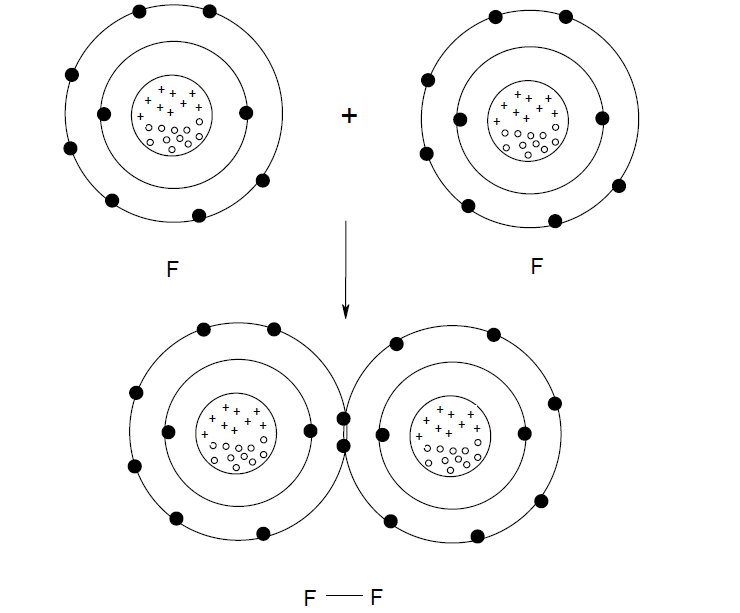
Figure 1. F-F covalent bonding.
A coordinate bond (also called a dative bond) is formed when an ion has a pair of electrons in the outer shell and uses that pair to join to another atom that has empty valence orbitals. The bond formed consists of lone pair sharing with the empty orbital of the second atom, as shown in Figure 2.
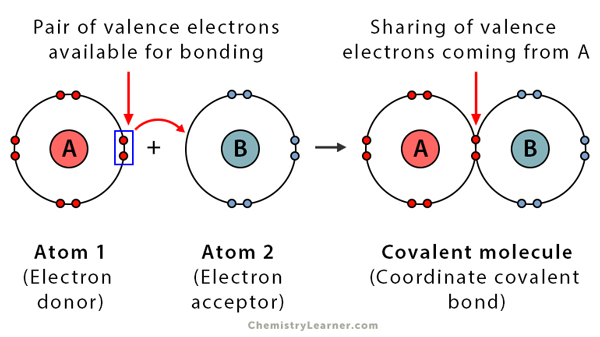
Figure 2. Example of a coordinate covalent bond.
Hydrogen bonding involves a hydrogen atom and is a weaker bond than an ionic or covalent bond but stronger than van der Waals bonding (see Figure 3). It involved a hydrogen atom bonded to an atom that has a high affinity for electrons. Hydrogen bonding can occur between atoms, within the same molecule, or in parts of the same molecule.
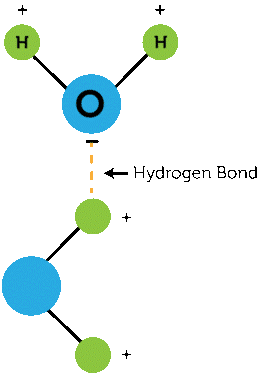
Figure 3. Example of a hydrogen bond.
Van der Waals forces or bonds are weak intermolecular interactions observed in solids and liquids responsible for bulk properties of substances such as boiling and melting points. These are secondary bonds, whereas covalent and ionic bonds are primary bonds. Figure 4 shows an example of van der Waals bonding where the atoms are polarized and attract one another.
Figure 4. Example of van der Waals bonding.
Coordination and hydrogen bonds can be formed between different units in the material, contributing to the overall strength of ion interactions in ionic exchange materials. In studies related to removing organic compounds from water, materials with a low number of charged groups are better at eliminating organics than ion exchange materials with many ion-exchange groups. This is due to the sorption of larger organic ions influenced by hydrogen and nonionic exchange interactions. In this case, the contribution of the weak forces overwhelms the contribution of the ion exchange itself. The van der Waals forces are responsible for the sorption in many large bio-organic molecules based upon studies using infrared spectroscopy.
Therefore, different types of forces may contribute simultaneously to interactions between large organic molecules and ion exchange materials. Various weak forces are often treated as independent and additive to the primary forces of covalent and ionic bonding; however, they may not be exclusive to these secondary forces. Also, other forces may be involved during these ionic interactions, but they are often considered small enough to be negligible. Examples of these forces are repulsion, collision mechanisms, actual particle morphology, and other interactions in the solution phase.
Effects of Ionic State
Many types of ions only convert to the ionic state when the pH of the solution or the ion exchange medium is at a certain level. When this occurs, the ion may be completely neutral at one pH and then convert to the ionic state once it is exposed to an acid or basic pH (recall Chemistry lessons on Brønsted-Lowry acid-base definition and percent ionization). This frequently happens with biochemical ions or when several groups of counterions are chargeable. Many ion exchange materials may be acidic or basic, so the conditions may dramatically change when the ion is transferred from the aqueous phase into the ion exchanger. An example is the sorption of phenol on OH- form of a weak-based resin at a pH of 5. Phenol is not charged at a pH of 5, but it is sorbed through the functional groups through the following reaction:
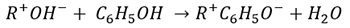
The phenol molecule converts to the ionic form when it is transferred to the ion exchanger phase. As the pH decreases below 5, other anions are sorbed by the exchanger and compete with phenol (Figure 5).
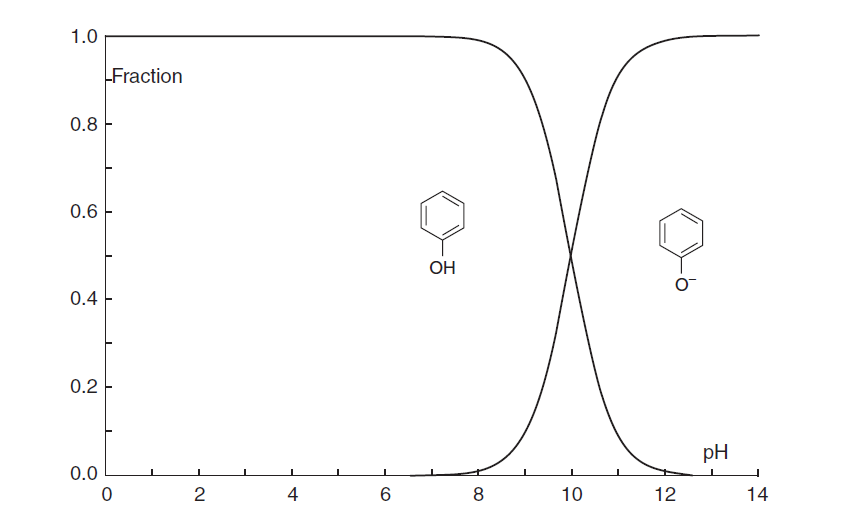
Figure 5. Ionization of phenol in aqueous solutions (phenol molar fraction vs. pH).
Certain substances have many functional groups and can have several different ionic forms. It isn't easy to study these systems because the interaction of these molecules with ion exchange materials contains ions in both phases at any pH. An example of a molecule that demonstrates this complexity is amino acids. Depending upon the conditions, the same amino acid can be cationic, anionic, or even zwitterionic because different groups of the same molecule can be charged oppositely. Some cases in the literature include mixtures of soluble polymers with no defined chemical structure with various functional groups. Studies on these systems are often empirical because it is too difficult to discern what is occurring, and therefore, has limited practical interest.
Positioning of the Functional Groups
The position of the functional groups in a molecule is significant for ionic interaction. The association of the ion exchange groups in the ion exchange material with the counterion results in the distortion of the matrix chains or the counterion steric structure.
The charged groups of large organic ions are often attached to flexible hydrocarbon chains and well-distanced. Due to this, the interaction of groups with functional groups of an ion exchanger does not cause too much strain on the polymer matrix. However, in general, the free energy changes due to the position of the functional groups. Therefore, the strength of the organic binding can be higher than the binding of the smaller ions due to the absence of counterions from the matrix elasticity force.
Hydrophobic Interactions and Complicated Hydration Effects
The hydration of the ion exchange material is affected by the hydrophobic and hydrophilic parts of the same molecules. Hydrophobicity is provided by functional groups such as –COOH, –OH, –NH3, =NH2, ≡N, =S. In organic molecules, hydrophobicity often comes from the hydrocarbon parts of a molecule. The water transfer through the ion exchange material is affected by the hydrophobicity, and we can express this relationship using the following equation:
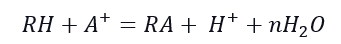
where n is not equal to zero. The literature also shows that hydrophobic interactions can dominate the sorption process; as the number of aromatic rings increases, the selectivity of the ion exchange resin increases. Therefore, the hydrophobic interaction plays an essential role in the selectivity of hydrophobic ions. However, there is no simple attraction between the hydrophobic matrix in the ion exchanger and the hydrophobic parts of the ions. The hydration structure reorganizes itself and is vital in the overall reaction. The sorption process may involve structured water clusters surrounding the hydrophobic areas and the expulsion of water molecules from the ion exchange material, increasing the overall entropy. The interaction between organic molecules and ionic exchange materials is typical for those matrices with ion-exchange groups. The presence of the ionic groups is essential for the swelling ability and the ion exchange process.
Size Effects
If the ion has a large size, it may not enter the dense ion-exchange network, which is termed "sieve action." Some larger molecules may enter the phase but may take a long time to interact due to the very slow diffusion. This phenomenon creates a need for low-crosslinked materials, even though this changes material characteristics. On the other hand, this characteristic can help scientists to design efficient separation processes. One method of achieving this consists of creating a chemical reaction to exchange small cations for a different group to enable fractionalization based upon molecular sizes.
An example of the "size effect" with ion exchange materials is Zeolite KX, which readily sorbs amino acids such as glycine, alanine, and lysine. However, their diffusion rates inside the ion exchanger can differ substantially. Larger amino acids such as phenylalanine cannot enter the zeolite phase, so separating amino acid types could be based on this concept.
Generally, gel resins with a low degree of cross-linking will remove organics from an aqueous solution more efficiently than highly cross-linked ion exchange materials. As mentioned in previous blog posts, the ion exchange removal efficiency varies with water content, regardless of the degree of cross-linking or polymer composition. Therefore, it is important to consider the linear relationship between water content and uptake of organics, irrespective of the actual chemical composition of the ion exchange system.
Conclusion
Ion exchange materials have many uses, and most manufacturers only discuss the ionic interactions that occur in the material. Many other factors are affecting the usefulness of the material, including water content and ion exchange capability. When ion exchange materials involve organic and biochemical molecules, other organic and biochemical complexities are involved with the interaction. There are secondary forces between the ionized group and counterion, such as coordination, van der Waals, hydrogen, and other molecular forces. Depending upon the counterions and ion exchange material, the position of the functional groups can affect ion transport, and steric hinderances can reduce ionic interactions. Also, the hydration properties may be complex depending upon the nature of the molecules and the hydrophobicity of the chains and functional groups. Finally, the pH of the ion exchange medium and the aqueous solution can affect the percent ionization, which means that minor pH adjustments may significantly affect the number of ionized species in the aqueous media and ion exchange medium.

 Posted by
Posted by









Enter the code in the box below: