One of the greatest challenges associated with PEMFCs is the water balance in the fuel cell stack. As the chemical reaction occurs in each cell, water is generated. Depending upon the load and the operating conditions, there is a tendency for the fuel cells to both flood and dry-out. The water content in the polymer membrane affects the proton conductivity and affects activation overpotentials. If the MEA is not adequately humidified, the protonic conductivity decreases, which means that the cell resistance increases.
Excess water can create an issue in fuel cells. It can prevent reactant diffusion to the catalyst sites by flooding of the electrodes, gas diffusion backings, or gas channels if the water removal is inefficient. For the membrane to remain hydrated, parameters such as current density, temperature, reactant flow rates, pressures, humidification, cell design, and component materials need to be optimized. Water in a PEM fuel cell arises due to the cathode reaction and the humidification of the reactant gases. The water production in the cathode depends upon the current density (the current being drawn by the load). Water buildup in a fuel cell can be mitigated through the gas diffusion layer, flow channel or heater design.
Water is transported mainly through the gas channels, but also through the membrane and the electrodes. Two transport processes that occur in the polymer membrane are diffusion and the electro-osmotic drag:
1. Electro-osmotic drag happens when hydrogen protons travel through the polymer membrane from the anode to the cathode and carry water molecules with them. The average number of water molecules “dragged” by a single proton is called the electro-osmotic drag coefficient.
2. Back diffusion occurs when the concentration gradient in the cathode drives the diffusion of water through the membrane.
The accumulation of water at the cathode occurs from both the electro-osmotic drag and the production of water at the cathode. The water at the cathode can be either transported to the flow channel through the gas diffusion backing, evaporated through heaters, or diffused through the membrane towards the anode.
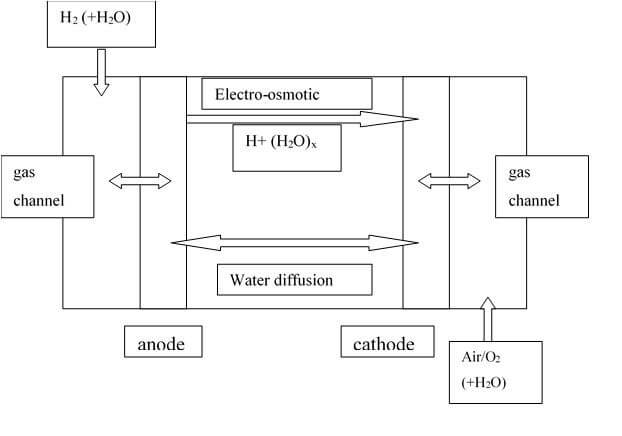
Figure 1: Water transport processes in a PEM fuel cell
Water can be removed from a PEM fuel cell is through the reactant streams. Undersaturated reactant gases pick up the liquid water in the flow channels. The water removal rate and the ratio of liquid and gaseous water depend upon the operating conditions and cell design. The operating temperature, pressure and flow rates have a substantial effect on water vapor content of the gases, water evaporation rate and vapor pressure. Therefore, the efficiency of water removal depends upon these parameters. The ratio of liquid and gaseous water is determined by the operating temperature and the humidity of input reactant gases.
When the product water produced at the cathode is not sufficient to keep the membrane hydrated, the use of humidified reactant gases is necessary. This occurs when the membrane becomes dehydrated due to high current density requirement at the anode. This phenomenon happens when the electro-osmotic drag is stronger than the water back-diffusion, which results in a greater flux of water from the anode to the cathode side. This problem is intensified when thick polymer membranes are used because they require more water to remain hydrated.
The humidification of the anode can be controlled through hydrogen humidification. Managing the water balance at the cathode is difficult because water accumulates due to the reaction and the electro-osmotic drag. The gas diffusion layers help to remove water from the electrodes, however, the water concentration gradient can depend upon the porosity, thickness and hydrophobicity of the gas diffusion backings. The flow channel geometry also influences the water content in the fuel cell. The width, length, depth and space between flow fields affect the flow velocity and rate of diffusion of the reactants to the electrode layers. It also determines how liquid water flows through the cell.
Depending upon the fuel cell application, the use of gas humidifier subsystems may not be desirable due to additional weight, size, system complexity, and parasitic power required. Besides humidifying the reactant gases, there are several methods of controlling water in the fuel cell:
• Direct water humidification of the MEA
• Thinner membranes to enhance water back diffusion
• Internal membrane humidification by embedded catalyst particles
• Improvement of the gas diffusion layers
For small, portable applications, the dead-ended bipolar design aids in preventing water accumulation. The electro-osmotic drag and water back diffusion is balanced; therefore, water accumulation in the anode does not occur.
Water management at the cathode is affected by the temperature, cathode flow, channel structure, air flow rate, and the gas diffusion backing. If air is the oxidant used in the fuel cell, it can be brought into the cell using natural convection or by using a small fan or blower. Natural convection is the simplest solution for small, portable fuel cells. Forced convection can make the fuel cell more efficient, but water management will be more difficult with this option.
Water balance must be part of the design consideration for a fuel cell stack. The correct balance of water can increase the efficiency and power output of the fuel cell and maintain the longevity of the fuel cell stack. The design parameters that should be considered for proper water balance include the overall stack dimensions, nominal power requirements, flow field design, GDL porosity & hydrophobicity, humidification levels, membrane thickness and any external plant components.

 Posted by
Posted by























Enter the code in the box below: