Electrolyzer 65
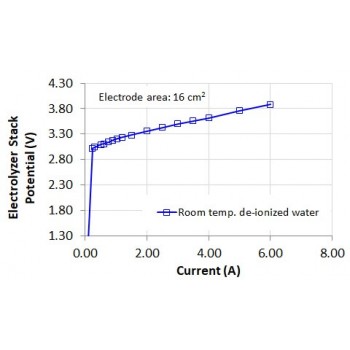
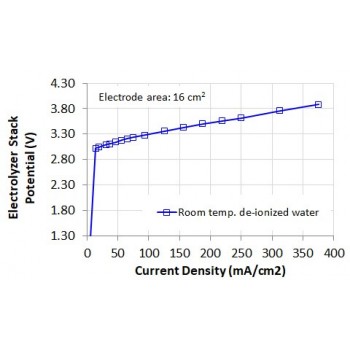
The Electrolyzer 65 is Double-Cell PEM electrolyzer stack, for production of hydrogen from distilled water. The center of the cell houses the hydrogen side of the two individual cells. The two outer sides of the cell have the function of supplying water and removing the oxygen produced. The two individual cells are connected electrically in series. Both cells have an electrode area of 16cm2.
The electrolyzer provided in this set is maintenance-free. However, always remember to use fresh, distilled water each time and to drain the water from the storage tanks after use.
• Hydrogen Production Rate: 65 cm3/min
• Oxygen Production Rate: 32.5 cm3/min
• Hydrogen Storage Volume: 80 cm3/min
• Oxygen Storage Volume: 40 cm3/min
• Power Consumption: 16 Watts at 4.0 VDC
• Permissible Operating Voltage: 0 - 4 VDC
• Permissible Operating Current: 0 - 4.4 A
• Permissible Operating Pressure: 0 - 5 mbar
• Electrode Area: 2 cells at 16cm2 each
• Operating Medium: Distilled Water
• Dimensions (H x W x D): 9.9" x 9.9" x 4.7" (250 x 250 x 120 mm)
• Weight: 2.1 pounds (950 g)
The growing significance of PEM electrolyzers mirrors the development of fuel cells. Electrolyzers generate the hydrogen required by fuel cells from water in an environmentally conscious manner. The electrical energy required for this purpose can be gained from renewable sources such as solar cells, wind farms or hydroelectric plants.
Water reacts in the electrolyzer under the influence of electrical energy according to the following formula: 2H2O = 2H2 + O2. This process takes place in the MEA (membrane electrode assembly). The MEA consists of the cathode, the anode, and a special polymer membrane (PEM) which is permeable to protons but which presents a barrier to electrons. Your h-tec electrolyzer functions on the PEM principle. The energy stored in chemical form in the gases can be converted back to electrical energy in a fuel cell as and when required.
Write a review
Your Name:
Your Review: Note: HTML is not translated!
Rating: Bad Good
Enter the code in the box below:










 H-TEC Education E206 Electrolyzer 65 Manual
H-TEC Education E206 Electrolyzer 65 Manual
























