Section 1

Section 1: Energy is a Physical Quantity That Follows Precise Natural Law
1.1 Energy is a quantity that is transferred from system to system. Energy is the ability of a system to do work. A system has done work if it has exerted a force on another system over some distance. When this happens, energy is transferred from one system to another. At least some of the energy is also transformed from one type to another during this process. One can keep track of how much energy transfers into or out of a system.
1.2 The energy of a system or object that results in its temperature is called thermal energy. When there is a net transfer of energy from one system to another, due to a difference in temperature, the energy transferred is called heat. Heat transfer happens in three ways: convection, conduction, and radiation. Like all energy transfer, heat transfer involves forces exerted over a distance at some level as systems interact.
1.3 Energy is neither created nor destroyed. The change in the total amount of energy in a system is always equal to the difference between the amount of energy transferred in and the amount transferred out. The total amount of energy in the universe is finite and constant.
1.4 Energy available to do useful work decreases as it is transferred from system to system. During all transfers of energy between two systems, some energy is lost to the surroundings. In a practical sense, this lost energy has been “used up,” even though it is still around somewhere. A more efficient system will lose less energy, up to a theoretical limit.
1.5 Energy comes in different forms and can be divided into categories. Forms of energy include light energy, elastic energy, chemical energy, and more. There are two categories that all energy falls into: kinetic and potential. Kinetic describes types of energy associated with motion. Potential describes energy possessed by an object or system due to its position relative to another object or system and forces between the two. Some forms of energy are part kinetic and part potential energy.
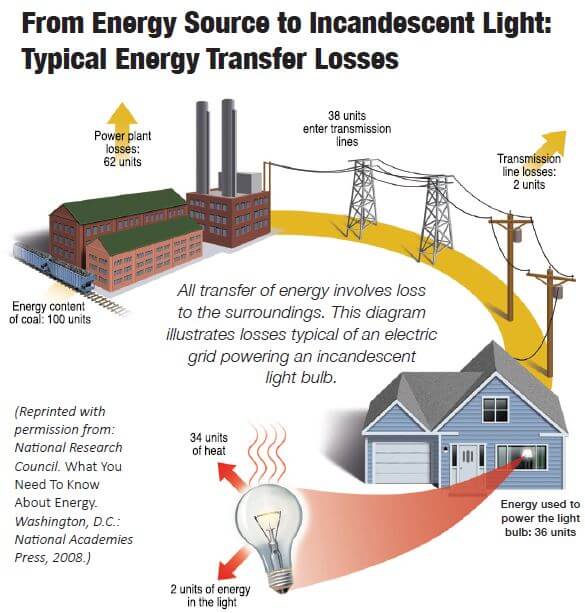
1.8 Power is a measure of energy transfer rate. It is useful to talk about the rate at which energy is transferred from one system to another (energy per time). This rate is called power. One joule of energy transferred in one second is called a Watt (i.e., 1 joule/second = 1 Watt). All transfer of energy involves loss to the surroundings. This diagram illustrates losses typical of an electric grid powering an incandescent light bulb.
1.6 Chemical and nuclear reactions involve the transfer and transformation of energy. The energy associated with nuclear reactions is much larger than that associated with chemical reactions for a given amount of mass. Nuclear reactions take place at the centers of stars, in nuclear bombs, and in both fission- and fusion based nuclear reactors. Chemical reactions are pervasive in both living and non-living Earth systems.
1.7 Many different units are used to quantify energy. As with other physical quantities, many different units are associated with energy. For example, joules, calories, ergs, kilowatt-hours, and BTUs are all units of energy. Given a quantity of energy in one set of units, one can always convert it to another (e.g., 1 calorie = 4.186 joules).









