Carbon Black - Ketjenblack EC-300J
Conductive blacks are classified as specialty carbon blacks. Ketjenblacks, as well as many other conductive blacks in the market, are furnace blacks. They are produced with the furnace process, where oil related streams undergo an incomplete combustion in an oven at drastic conditions.
Ketjenblack itself is a material with an intrinsic resistivity of about 0.01 - 0.1 [Ω·cm]. Ketjenblack is mostly available as pellets of around 3 mm (Figure 1). These pellets can be easily broken into the smaller particles that constitute them: the agglomerates, with a size of roughly 100 micrometers (μm). Agglomerates are formed by several aggregates, which are even smaller particles of roughly 0.1 - 1 μm. Aggregates are the key particles for conductivity. They tend to stick together, and high shear forces are necessary to break the agglomerates into aggregates.
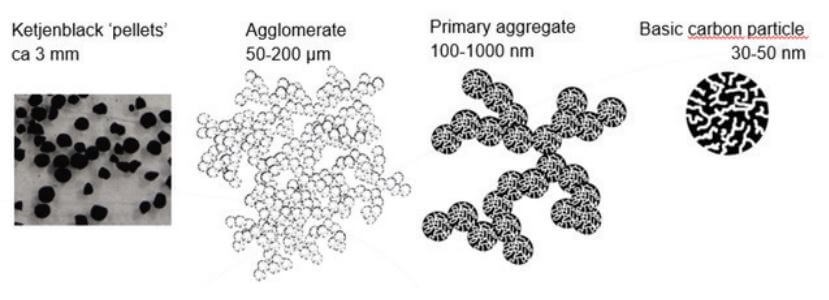
Figure 1. Ketjenblack Structure
To get conductivity, the carbon black particles need to form a network through which the electrons can be transferred. The percolation curve shows the formation of a conductive network (Figure 2). Until a certain dosage, no much change in conductivity (the inverse of resistivity) occurs. At a certain critical concentration, the polymer/rubber starts becoming conductive (percolation point).
When the percolation curve becomes flat, addition of more conductive black will not change the conductivity. Before the percolation curve, there is just not enough conductive filler: the distance between the carbon black particles is too long and electrons cannot transfer from one particle to the other. When percolation is reached, the distance is just enough to allow electron transfer.
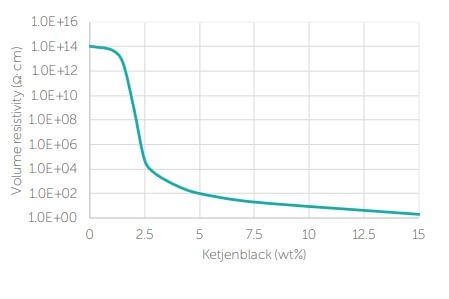
Figure 2. Percolation curve
During mixing or compounding, Ketjenblack must first be wetted by the polymer in the compound matrix, which takes time, due to its high surface area and pore volume (low bulk density). The wetting process can be controlled regulating the mixing temperature. Furthermore, sufficient mixing-energy must be provided to disperse the wetted carbon black uniformly into the compound.
Essential for such a uniform distribution of carbon black is enough shear during compounding. The regulation of temperature (wetting) and shear forces (dispersion of Ketjenblack agglomerates) are two very important parameters that need to be carefully calibrated to find the best compromise, especially considering the type of polymer in use.
Write a review
Your Name:
Your Review: Note: HTML is not translated!
Rating: Bad Good
Enter the code in the box below: