When a PV cell is exposed to sunlight, the photons of the absorbed sunlight dislodge the electrons from the atoms of the cell. The free electrons then move through the cell, creating and filling in holes. It is this movement of electrons and holes that generate electricity. The process of converting sunlight into electricity is known as the “photovoltaic effect.” Light is a form of energy, and electrons begin to move when light energy enters the material. The electrons freely flow through the crystalline structure and are collected using electrically conductive metals such as copper. The accumulated electrons produce the current and the cell voltage (power) output of a solar cell.
Many PV cells are connected to form modules, which are then assembled into larger units called arrays to increase the power output. These units of PV cells allow designers to build PV systems with customized power output for different applications. A complete PV system consists of PV modules, support structures, wiring, storage, power conversion, and power electronics devices.
A silicon atom has 14 electrons arranged in three different shells. The first two shells are full, and the third shell is partially empty, having only four electrons as shown in Figure 1. An atom in its ideal state needs eight electrons in its last shell. Therefore, to compensate for the empty spaces, it shares four electrons with its neighboring silicon atoms (this is what forms the crystalline structure). The material properties of silicon in its “pure” state allows it to be a conductor because its electrons are unable to move around (they are locked in this crystalline structure).
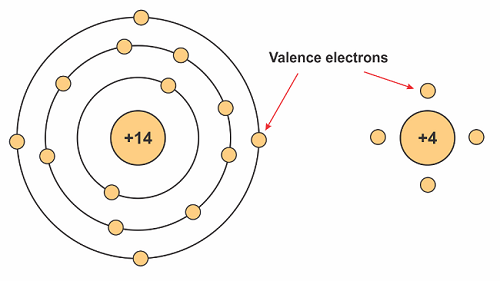
Figure 1. Silicon has 14 protons and electrons, but atoms are typically written with their valence electrons only.
Therefore, the silicon material properties need to be modified to allow the electrons to move around. This task is accomplished by implanting impurities into the silicon material. When other types of atoms are mixed into the material, this enables the electrons to move. For example, a phosphorous atom has five electrons in its outer shell, and if these are placed into the silicon material, it still bonds with the silicon atoms, but there is one free electron that is not bonded.
When a photon hits a piece of silicon, it can either be absorbed by the silicon, reflect off of the surface, or pass through the silicon. The photon path depends on whether the photon energy is higher or lower than the band gap. If the photon is absorbed, its energy is given to an electron in the crystal lattice. The valence electron is usually bonded tightly due to neighboring atoms; however, the additional energy provided by the photon excites it into the conduction band, where it can move around in the semiconductor. The electron moves to another location (hole) and leaves a “hole” where it once was. This phenomenon is called mobile “electron-hole” pairs in the semiconductor.
The electromagnetic spectrum is comprised of many different wavelengths. Optical radiation is thought of as "light," but optical or "visible" light is just a tiny part of a much broader range of energy. Many other portions of the spectrum are totally or partially blocked by earth's atmosphere. The only way to observe the parts of the spectrum is to go into space! The names of the various sections of the electromagnetic spectrum are shown in Figure 2. When you witness a rainbow after a storm, you observe the separation of light into its different wavelengths. Since PV materials only absorb a certain range of energies, certain wavelengths of light will be unable to create free electrons. Depending upon the material type, a certain amount of energy (1.1 eV for crystalline silicon) is required to make electrons move; this is known as the “the band gap energy” of the material.
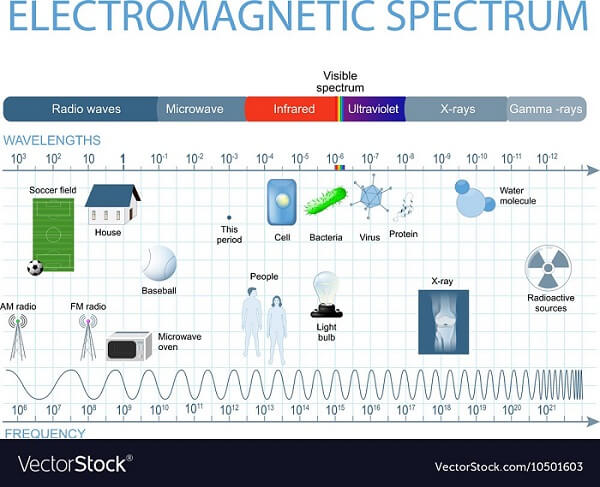
Figure 2. The electromagnetic spectrum.
The band gap also determines the strength (voltage) of the electric field. If it is too low, the extra absorbed photons do not create a high enough voltage to produce the required power. There are also electron losses due to the contact area between the metal and material. Theoretically, the larger the metal contact, the more electrons it can collect. However, a significant portion of the material cannot be covered because light needs to enter the material to generate the electric current. Also, silicon is a semiconductor (not a conductor), which means that it is difficult for electrons to travel through the material due to the high internal resistance. The optimal band gap is 1.4 eV for a cell material to balance these two effects.
When energy is added to pure silicon, a few electrons break free from the lattice – which leaves a “hole.” These free electrons then attempt to become stable by looking for another hole. These free electrons are termed “free carriers.” The silicon with the extra phosphorous electrons (doped) allows enough electrons to move to be able to conduct current. The process of adding electrons is called “doping,” and the silicon materials with the phosphorous atoms is termed “n-type silicon.” Silicon material can also be doped with boron, which has only three electrons in its outer shell compared with the four that silicon has. This type of silicon is called “p-type,” and therefore, silicon has free “holes” instead of electrons. These “holes” move around like the electrons and carry a positive charge. If p-type silicon is placed into contact with n-type silicon, then the electrons will diffuse from a region of high concentration (n-type side) to a region of low concentration (p-type side). The electrons in the n-type material are repelled by the negative electrode and are drawn to the p-type electrode. The holes in the p-type material move the opposite way.
When the difference in voltage between the electrodes is high, the electrons in the depletion zone come out of their holes and begin moving freely again. The depletion zone disappears, and the charge moves across the diode. Figure 3 illustrates the P-N junction before and after the electrons begin to move.
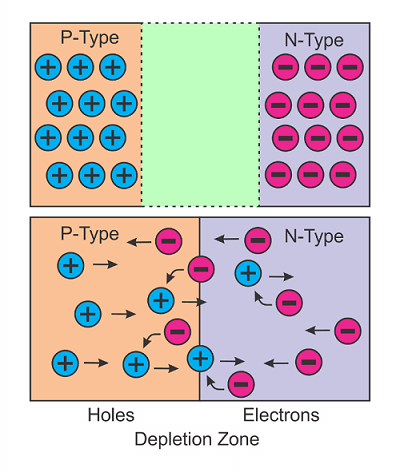
Figure 3. P-N junctions.
Metal contacts are placed onto the n-type and p-type sides of the solar cell, and the electrodes are then connected to the device that needs to be powered. The electrons move from the n-type side to power the load and then travel to the P-type semiconductor-metal contact. They then recombine with a hole that was created by an electron-hole pair on the P-type side or are swept across the junction from the N-type side after being created there. Figures 4 illustrates the concept as current flows across the junction.
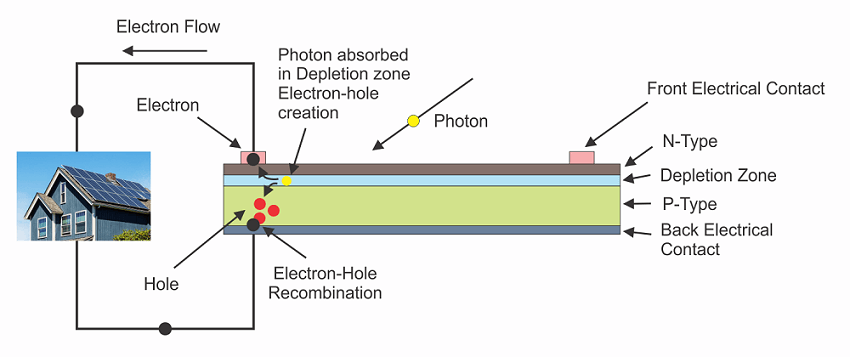
Figure 4. Solar cell connected to an external load.
Although silicon is the most common type of material used for PV cells, other materials can also be used. Polycrystalline silicon is sometimes used to reduce the manufacturing costs – although solar cells made of polycrystalline are not as efficient. Amorphous silicon can also be used to reduce production costs. Other common materials include:
1. Gallium Arsenide
2. Copper Indium Diselenide
3. Cadmium Telluride
Each type of material has a different “band gap,” which means that it absorbs different wavelengths of energy. One method of improving PV efficiency is to use two or more different material layers. These are called “multi-junction” cells and can absorb different wavelengths of energy. The higher band gap is usually on the surface absorbing the high energy photons, and the lower band gap material is underneath. Figure 5 shows the portion of the periodic table that is relevant to photovoltaics.
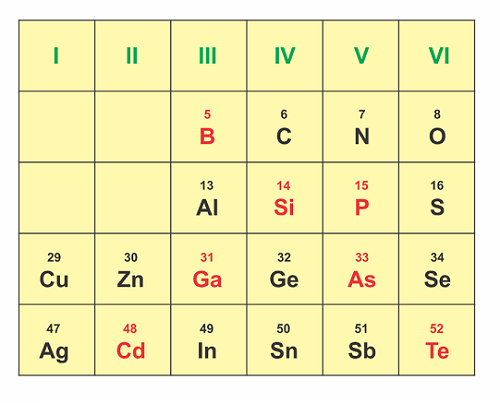
Figure 5. The portion of the periodic table relevant to photovoltaics.
The material most frequently used for PV panels is crystalline silicon, which has an efficiency of approximately 15%. It can be made from a silicon ingot, ribbon, or wafer. Monocrystalline silicon is usually formed using the Czochralski process. These panels are expensive because they are made of pure cylindrical ingots. Figure 6 shows the basic steps in the forming solar modules.

Figure 6. Material steps in forming solar modules.
Cadmium telluride (CdTe) is another efficient light-absorbing material for solar cells. CdTe is easy to manufacture and suitable for large-scale production, and it is the only technology besides amorphous silicon that can be manufactured on a large scale. However, there is a perception of toxicity with CdTe solar cells because they are based upon cadmium, which is a metal that can act as a cumulative poison. Many studies have shown that the release of cadmium in the cadmium telluride solar cells is lower than in other cadmium-based solar cell technologies.
Copper-Indium Selenide (CuInSe2) solar cells have high light absorption properties. The optical and electrical characteristics can be manipulated to build customized devices. Some of the films have achieved greater than a 14% efficiency; however, the manufacturing costs are high in comparison with silicon solar cells.
Gallium arsenide multijunction cells are high-efficiency cells that were initially developed for specialized applications such as satellites and space exploration. These cells consist of several layers, such as GaAs, Ge, GaInP2 and are deposited using metalorganic vapor phase epitaxy.
The semiconductors that are chosen to make up these multijunction cells are chosen to absorb nearly the entire solar spectrum, therefore, generating the maximum amount of energy. GaAs-based multijunction solar cells are the most efficient solar cells with an efficiency of approximately 40%. This technology is currently being utilized in the Mars rover missions.
Light absorbing dyes (DSSC) are currently being investigated for solar cell technology. An example is a ruthenium metallorganic dye that is deposited in a thin layer to absorb light. The dye-sensitized solar cell depends upon a layer of nanoparticulate titanium dioxide (TiO2) to maximize the surface area (200 – 300 m2/g TiO2, compared with 10 m2/g of the surface area of the crystal). When light hits this solar cell, the electrons are passed to the n-type TiO2, and the holes move to the electrolyte on the other side of the dye. This cell allows flexibility in the materials that are chosen and uses low-cost manufacturing methods such as screen printing. However, the dyes suffer from heat and UV light degradation, and the casing can be difficult to seal due to the solvents used in the assembly.
There are many organic and polymer solar cells that are fabricated from thin films (~ 100 nanometers) and are currently investigated for solar cells. Examples include polymers, thin films, and specialty materials. Energy conversion efficiencies are presently much lower than other solar cells types with the highest efficiency reported at 6.5%. This cell type would still be valuable where mechanical flexibility is essential.
Photovoltaic cells can conduct electrons because they use semiconductor materials. Silicon is the most common type of semiconductor material used for PV cells. A few other materials used are gallium arsenide, copper indium diselenide, and cadmium telluride. Solar systems will be an essential part of the future hybrid energy system due to its proven ability to use solar energy to produce power. Since solar energy can be intermittent, it would ideally be used with a system consisting of batteries, an electrolyzer, fuel cell, or wind power.

 Posted by
Posted by




















