 A biological fuel cell (BFC) or microbial fuel cell (MFC) is a type of fuel cell that converts biochemical energy into electrical energy. Like other types of fuel cells, a biological fuel cell consists of an anode, a cathode, and a membrane that conducts ions. In the anode compartment, fuel is oxidized by microorganisms, and the result is protons and electrons. In the cathode compartment, ions are consumed, and the by-product is water. In BFCs, there is the redox reaction between the carbohydrate substrate (such as glucose and methanol) and the catalyst -- which is a microorganism or enzyme. The biological fuel cell is illustrated in Figure 1. The main difference between a standard fuel cell and a BFC is the catalyst is a microorganism or enzyme. Therefore, noble metals are not needed for the catalyst in BFCs. The fuel cell operates in a liquid media in a near neutral environment and at a low temperature. The potential applications for biological fuel cells are (1) low power energy sources; (2) sensors based upon direct electrode interactions; and (3) electrochemical synthesis of chemicals.
A biological fuel cell (BFC) or microbial fuel cell (MFC) is a type of fuel cell that converts biochemical energy into electrical energy. Like other types of fuel cells, a biological fuel cell consists of an anode, a cathode, and a membrane that conducts ions. In the anode compartment, fuel is oxidized by microorganisms, and the result is protons and electrons. In the cathode compartment, ions are consumed, and the by-product is water. In BFCs, there is the redox reaction between the carbohydrate substrate (such as glucose and methanol) and the catalyst -- which is a microorganism or enzyme. The biological fuel cell is illustrated in Figure 1. The main difference between a standard fuel cell and a BFC is the catalyst is a microorganism or enzyme. Therefore, noble metals are not needed for the catalyst in BFCs. The fuel cell operates in a liquid media in a near neutral environment and at a low temperature. The potential applications for biological fuel cells are (1) low power energy sources; (2) sensors based upon direct electrode interactions; and (3) electrochemical synthesis of chemicals.
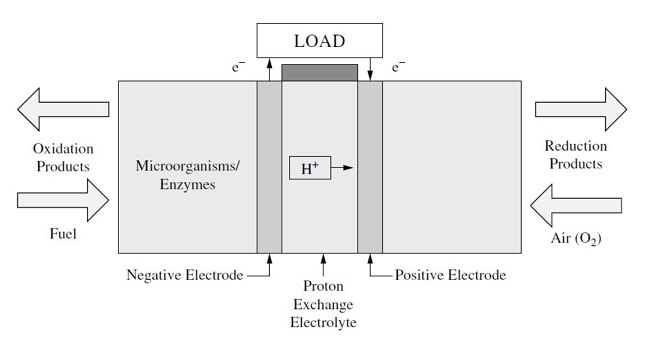
Figure 1: A biological fuel cell (BFC)
During the last decade, improvements have been made in the selection of the microorganisms, process kinetics, and the use of different mediators to improve electron transfer. Also, many types of electrodes have been studied for reaction efficiency.
In bacteria cells, mitochondria serve as the energy storage unit by accumulating or releasing chemical energy in the form of substances like nicotinamide adenine dinucleotide with high-energy hydrogen (NADH) or nicotinamide adenosine dinucleotide phosphate (NADPH). The NADH and NADPH act as electron transfer paths from the substrate to the metabolites. The NADH/NAD ratio increases as the oxygen limitation becomes more severe.
Most of the substrate is converted into an electroactive substance through control of bacteria metabolism. However, the biological reactions mentioned earlier only take place in diluted aqueous media -- which is not always suitable for charge-transfer reactions. The electron transfer from these electroactive substances to the electrode is a slow process. Therefore, a suitable redox mediator is needed to improve the electron transfer and the electrode reaction.
In a study done by the Helinski University of Toronto, a mediator 2-hydroxy-1,4-naphtho-quinone (HNQ) was used to improve the electron transfer. When this mediator substance is present in the system, the color in the biofilm reactor changes. The changes in the color occurs because of the metabolic activity of the biofilm reactor and the electron transfer in the fuel cell anode. Thus, it offers a simple method of monitoring and controlling functions of the fuel cell.
Experimental tests of the bacterial fuel cells have resulted in several conclusions. First, the conversion rate of electrical energy is lower (15 to 25 percent) than chemical fuel cells due to the complex reactions. Secondly, the current density per anode volume increases when the size of the fuel cell decreases. Finally, the power output increases if the bacteria are immobilized. Another similar type of fuel cell that has been investigated is the enzymatic fuel cell. Replacing the bacteria with an enzyme could potentially make the process easier to control because enzymatic reactions are simpler. According to researchers at the Helinski University of Technology, the conversion rate of the Enzymatic Fuel Cell (EFC) is expected to be greater than 50 percent. Experiments have shown that the rate is in the range of 40 to 55 percent for the bacterial fuel cell when the substrate is glucose. Similarly, the current density of the EFC is likely to improve significantly compared to that of the bacterial fuel cell.
In addition to using bacteria or enzymes to create energy in BFCs, there are also different ways to produce hydrogen through biological methods. Three main categories of hydrogen creation through biological processes are:
• Photosynthesis using unicellular organisms that use hydrogenase or nitrogenase reactions
• Bacteria that produce hydrogen anaerobically
• Processes that use a combination of bacteria to break down complex organic molecules into a compound that can be transformed to hydrogen using hydrogen-producing organisms
We will briefly introduce the photosynthesis and digestion processes that are currently under development.
Photosynthesis consists of the conversion of light energy to biochemical energy through a photochemical reaction and reduction of atmospheric carbon dioxide to organic compounds such as sugars. Certain plants and algae absorb solar energy, and several types of algae and cyanobacteria produce hydrogen instead of sugars during photosynthesis. These organisms have hydrogenase or nitrogenase enzymes to catalyze hydrogen formation.
Several types of green algae produce hydrogen. Some common ones that have been investigated are Scenedesmus, Chlamydomonas, C. reinhardtii, Anabaena Cylindrica, Synechococcus, Rhodobacter sp, Rubrivivax gelatinosus, Rhodovulum sulfidophilum, R. sphaeroides as well as many others. Scenedesmus produces hydrogen on light exposure after being kept in the dark under anaerobic conditions. This green alga is a “water splitting” organism, which means that it performs biophotolysis using hydrogenase to reduce water to hydrogen. Additional research work has been conducted with a mutant of Chlamydomonas to produce a continuous flow reactor using this type of algae. Although high efficiencies of light: hydrogen have been obtained, the hydrogenase reaction is very sensitive to oxygen, which has prohibited advances in development with this type of bacteria. New methods have been developed by the National Renewable Energy Laboratory (NREL) to improve hydrogen by controlling oxygen and sulfur in the environment of C. reinhardtii. The algae culture is grown under normal conditions and then deprived of oxygen and sulfur, which makes it switch to an alternate metabolism to produce hydrogen. This cycle can be continuously repeated to generate hydrogen.
A nitrogen-fixing cyanobacterium, Anabaena Cylindrica, produces hydrogen and oxygen simultaneously in an argon atmosphere for several hours. Hydrogen production occurs under normal atmospheric conditions, but at a much slower rate than nitrogen fixation. The hydrogen production can be increased by starving the algae of nitrogen.
Most of the photosynthetic systems being studied have low efficiencies. However, recent advances in genetic engineering may help create more efficient systems. One example is R. Sphaeroides, which has a photoenergy conversion of 7 percent, which seems low; however, it is significant considering the efficiency of photovoltaic systems is 10 percent.
Hydrogen can also be produced by the microbial digestion of organic matter in the absence of light. Many of these bacteria types produce hydrogen with low molecular weight acids. Unfortunately, the reaction rates are low, and a lot of hydrogen is not generated due to the inhibition of microbial hydrogenases, along with the reaction of hydrogen with other species. Hydrogen typically reacts with organic species or carbon dioxide, which produces methane. Thus, one of the main challenges in digestion processes is inhibiting methane production to favor hydrogen production. Increasing the temperature in these types of systems is not an effective method of improving the process efficiency because this usually denatures enzymes and bacteria. Some of the most promising bacteria include Escherichia coli, Aerobacter aerogenes, Aerobactin cloacae, Pseudomonas sp, and Clostridium butyricum. Clostridium butyricum has yielded 35 mmol (784 ml) of hydrogen per hour from 1 g of microorganism at 37 ºC. The use of biological processes for hydrogen production is at the point of technical system development, and the photosynthesis-algae-bacteria systems currently seem to be the most promising.
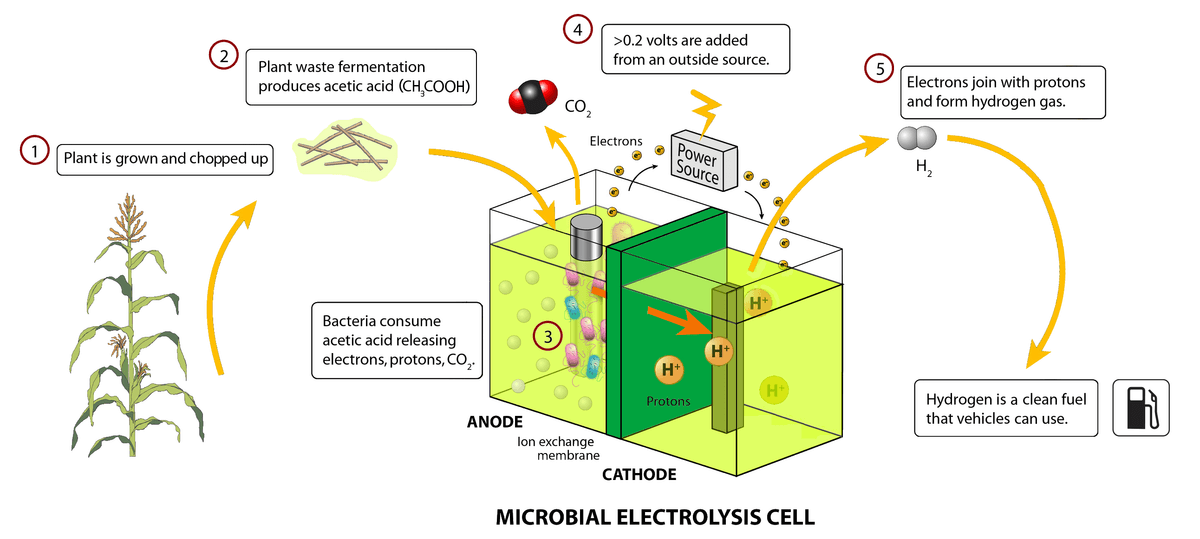
Biological power systems have many advantages over traditional chemical systems due to low-temperature operation and non-hazardous materials. These systems have the potential to eliminate the transport and storage of large quantities of hydrogen because the hydrogen is created in-situ. Also, the BFC eliminates the occurrence of catalyst poisoning over time due to the use of biological catalysts. These naturally-occurring systems hold the future of energy production for portable and stationary systems.

 Posted by
Posted by




















Enter the code in the box below: