 Each component of the fuel cell must be designed properly – otherwise, you run the risk of decreasing fuel cell performance. The bipolar plates are termed “bipolar” because they have flow fields on both sides. This design is very convenient when you have membrane electrode assemblies (MEAs) on both sides. In a fuel cell with a single cell, there are no bipolar plates because there is only one MEA. However, in fuel cells with more than one cell, there is usually at least one bipolar plate. Bipolar plates perform several roles simultaneously in fuel cells. Bipolar plates have reactant flow channels on both sides, forming the anode and cathode compartments of the unit cells on the opposing sides of the bipolar plate. They not only distribute fuel and oxidant within the cell, but they also separate the individual cells in the stack, collect the current, carry water away from each cell, humidify gases, and keep the cells cool. To simultaneously perform these functions -- specific plate materials and designs are needed.
Each component of the fuel cell must be designed properly – otherwise, you run the risk of decreasing fuel cell performance. The bipolar plates are termed “bipolar” because they have flow fields on both sides. This design is very convenient when you have membrane electrode assemblies (MEAs) on both sides. In a fuel cell with a single cell, there are no bipolar plates because there is only one MEA. However, in fuel cells with more than one cell, there is usually at least one bipolar plate. Bipolar plates perform several roles simultaneously in fuel cells. Bipolar plates have reactant flow channels on both sides, forming the anode and cathode compartments of the unit cells on the opposing sides of the bipolar plate. They not only distribute fuel and oxidant within the cell, but they also separate the individual cells in the stack, collect the current, carry water away from each cell, humidify gases, and keep the cells cool. To simultaneously perform these functions -- specific plate materials and designs are needed.
The flow field design needs to effectively distribute gases and liquids over as much of the surface area of the MEA as possible. Commonly used designs include straight, serpentine, parallel, inter-digitated or pin-type flow fields. Along with these designs, the plate materials are chosen based upon chemical compatibility, resistance to corrosion, cost, density, electronic conductivity, gas diffusivity/impermeability, manufacturability, stack volume/kW, material strength, and thermal conductivity. Therefore, you can see that the selection of a seemingly simple part of the fuel cell may take quite a bit of planning and design!

The design of the flow fields, along with the channel dimensions influence reactant flow velocities and mass transfer, and therefore fuel cell performance. Bipolar plate materials must have high conductivity and be impermeable to gases. Due to the presence of reactant gases and catalysts, the material should be corrosion-resistant and chemically inert.
Fuel cell bipolar plates account for most of the stack weight and volume in a fuel cell stack; therefore, it is desirable to produce plates with the smallest dimensions possible (< 3 mm width) for portable and automotive fuel cells. When flow channels are formed on both sides of the same plate, one side serves as the anode plate, and the other side is the cathode plate.
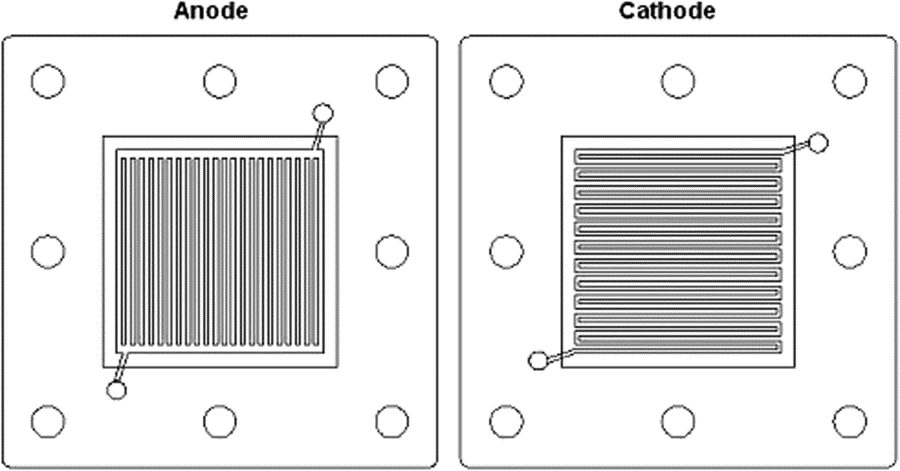
Most PEMFC and DMFC bipolar plates are made of graphite or resin-impregnated graphite. Solid graphite is highly conductive, chemically inert, and resistant to corrosion; but expensive and costly to manufacture so it is often used for prototypes. Flow channels are usually machined or electrochemically etched for prototypes or low-volume fuel cell stacks. These methods are not suitable for mass production, which is why alternative bipolar materials and manufacturing processes have been extensively researched for two decades.
Many types of metallic plates are suitable for fuel cells, including aluminum, steel, titanium, and nickel. Metallic plates are suitable for mass production and can be made into very thin layers, which results in lightweight and portable stacks. The bipolar plates are exposed to a corrosive environment, which means that dissolved metal ions can diffuse into the membrane, which lowers ionic conductivity and reduces fuel cell life. To prevent the metal migration, a layer is needed to prevent corrosion while promoting conductivity. Some commonly used layers are graphite, gold, silver, palladium, platinum, carbon, conductive polymer, and other types.
Graphite-carbon composite plates have been created using thermoplastics or thermosets with conductive fillers. These materials are chemically stable in fuel cells and are suitable for mass production techniques, such as compression molding, transfer molding, or injection molding. Often, the construction and design of these plates are a trade-off between manufacturability and functional properties.
Another important characteristic of bipolar plate materials is the electrical conductivity. The loss of conductivity in the cell is primarily due to contact resistance. This type of loss results from interfacial contacts between the bipolar plate and the gas diffusion layer. The interfacial contact resistivity losses can be determined by sandwiching a bipolar plate between two gas diffusion layers, and then passing an electrical current through the sandwich and measuring the voltage drop. The total voltage drop is a strong function of clamping pressure. Bulk resistance of the bipolar plate and the gas diffusion media should be independent of the clamping force, but the contact resistance is a strong function of the clamping force.
The most common metallic materials used for bipolar plates are aluminum, stainless steel, titanium, and nickel. Most metallic plates are made of one metal, but there are designs in the literature that use modular metallic bipolar plates, which combine different materials to enhance thermal and electrical conductivity. Bipolar plates need to be chemically resistant because corrosion or dissolution may occur depending upon the material. The formation of corrosion layers on the surface of the plate increases the electrical resistance. Also, depending upon the type of metal, ions can diffuse into the membrane and are trapped at ion exchange sites, thus lowering the ionic conductivity. Due to these issues, metallic bipolar plates use protective coatings.
Coatings for bipolar plates should be corrosion-resistant and protect the substrate from the operating environment. There are many carbon-based and metallic bipolar plate coating materials that can be used. Carbon-based coatings include graphite, conductive polymer, and organic self-assembled monopolymers, while metal-based coatings consist of noble metals, metal nitrides, and metal carbides.
Many methods are used for depositing coatings onto metallic bipolar plates. Processes include physical vapor deposition techniques like electron beam evaporation, sputtering and glow discharge decomposition, the chemical vapor deposition technique, and liquid phase chemical techniques like electro- and electroless deposition, chemical anodization/oxidation overcoating, and painting.
When selecting a coating, important considerations include conductivity, corrosion resistance, thermal expansion, and the absence of micropores and microcracks. Metal plates are exposed to temperature differentials in the fuel cell stack; therefore, the temperature range should be considered when selecting the coating and metallic plate type because the expansion and contraction maybe at different rates. The coating must adequately cover the plate and not be exposed to conditions that could cause microcracks or other coating defects. Micropores and microcracks may lead to failure if the base metal becomes exposed to the acidic fuel cell environment. Thermal expansion differences, as well as microcracks and micropores, can be minimized by adding intermediate coating layers between that of adjacent layers.
Many new fuel cell materials have been under investigation for the last couple decades. Many of these new plates are metal or carbon-based. There are several interesting material innovations, such as materials that combine porous graphite, polycarbonate plastic, and stainless steel to utilize the beneficial properties of each material type. In this example, the stainless steel provides rigidity to the structure while the graphite resists corrosion. The polycarbonate or other polymer type provides chemical resistance and can be molded to any shape to double-up as gaskets and manifolds. Some of these layered plates seem to be a good alternative from a chemical, physical, electrical, manufacturing and cost-perspective.
An extensive amount of research has been conducted on the use of carbon-based composite bipolar plates in fuel cells. Some of the carbon composite bipolar plates have been made using thermoplastic (polypropylene, polyethylene, and poly[vinylidene fluoride]) or thermosetting resins (phenolics, epoxies, and vinyl esters) with fillers and fiber reinforcement. Recent bipolar plate manufacturers are producing bipolar plates with thermoplastic resins because of the ease of manufacturability -- they are easily injection-moldable and recyclable.
The selection of the appropriate bipolar plates can be a challenge due to the required multifunctionality of the plates. The materials typically used for bipolar plates are graphite, coated metallic plates, and composite plates. The flow field design is also crucial for ensuring even distribution of the reactants and products throughout the cell. Many process options are available for manufacturing the bipolar plates. The options selected should be carefully considered to ensure it will both perform the optimally and will enable mass production of the fuel cell type.

 Posted by
Posted by




















Enter the code in the box below: