Article Search
SearchFuel Cell Store Blog
Fuel cell modeling is helpful for fuel cell developers because it can lead to fuel cell design improvements, as well as cheaper, better, and more efficient fuel cells. The model must be robust and accurate and be able to provide solutions to fuel cell problems quickly. A good model should predict fuel cell performance under a wide range of...
The performance of a fuel cell stack can be estimated using a few equations combined with some input data. A common way of characterizing performance of different fuel cell stacks is using polarization curves. Although you cannot pinpoint specific issues with these curves, they will allow you to calculate the overall performance. An example polarization curve is...
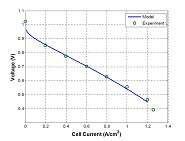
Model validation is the most important step in the model building process; however, it is often neglected. Even when the model is validated, it is often not done adequately. It often consists of taking a few experimental data points and plotting these points on the same graph as the model. There are two different types of models: engineering or...
As fuel cell size decreases, the transport phenomena of the fuels and water changes in the fuel cell. In standard fuel cell designs, the movement of fuel and water is governed by volumetric effects, but surface effects become critical as dimensions shrink. A good rule of thumb is that millimeter-scale devices are small enough for...
Small plant components are required to deliver the reactants to the fuel cell with the required conditions. Examples of these components are blowers, compressors, pumps, and humidification systems used to deliver the gases to the fuel cell with the proper temperature, humidity, flow rate and...
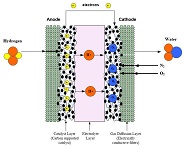
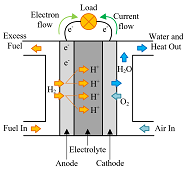
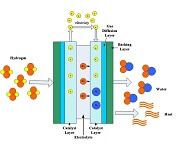
The fuel cell electrode layer is made up of the catalyst and porous gas diffusion layer. When the fuel in the flow channels meets the electrode layer, it diffuses into the porous electrode. The reactant travels to the catalyst layer where it is broken into protons and electrons. The electrons move to the...

The electrolyte layer is essential for a fuel cell to work properly. In PEM fuel cells (PEMFCs), the fuel travels to the catalyst layer and gets broken into protons (H+) and electrons. The electrons travel to the external circuit to power the load, and the hydrogen protons travel through the electrolyte until it reaches the cathode to combine with oxygen to form...

After the membrane electrode assembly (MEA) has been fabricated, it must be integrated into a fuel cell stack. The stack has multiple jobs, including evenly distributing fuel and oxidant to the cells, collecting the current to power the desired devices, and evenly distributing or discarding heat and...

Fuel cells are not limited to pure hydrogen gas as fuel. Each type of fuel cell stack has different fuel tolerances. The lower the operating temperature of the stack, the stricter the requirements for pure fuel. For fuels other than pure hydrogen, an external fuel processing system may...

Fuel cells often use compressed hydrogen as the fuel; however, many other hydrogen sources can be used with fuel cells. Chemical hydride storage is an alternative method of producing hydrogen via a chemical reaction. These reactions involve chemical hydrides, water, and alcohols. The chemical reactions are not reversible, and the byproducts must be discarded. Hydrogen fuel can also...

Fuel cells usually use compressed hydrogen as the fuel, but there are many other types of fuels that can be used. The type of fuel used depends upon the fuel cell application. Fuels are often in their final form before entering the fuel cell; however, certain fuel cell types can be processed on the inside of the fuel cell. Alternative fuel types are...
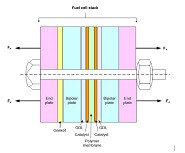
There are many steps involved in the manufacturing of a fuel cell stack. One of these steps is the hot pressing of the polymer electrolyte membrane to the two gas diffusion layers (GDLs). This creates a three-layer laminate membrane electrode assembly (MEA). Other steps involve the machining or etching of the...

This post will help you to determine the best location for a photovoltaic (PV) system. After you have sized your PV system based upon the calculated the power requirements, you will have to select a location that has maximum sun exposure and limited shading throughout the year. PV arrays can be...

When you receive your energy bill each month, you may not understand exactly how the total amount has been calculated. Every device or appliance in your household contributes to the total sum of the bill. To figure out which appliances and devices are using the most energy, you can estimate the...